Activity
Mon
Wed
Fri
Sun
Jul
Aug
Sep
Oct
Nov
Dec
Jan
Feb
Mar
Apr
May
What is this?
Less
More
Owned by Rowan
All-in-one peptide education community. 🧪 Dosing guides, storage protocols, stacking frameworks, vendor intel, GLP-1 research, and expert support.🧬
Memberships
Heather Dawn Research Wellness
3.5k members • Free
DadBod2Fit - SARM and Peptide
210 members • Free
Peptides & Pump Knowledge Base
1.8k members • Free
Research Peptide Community
1.7k members • Free
Peptides & Biohacking Hub
1.5k members • Free
The Iron Forge Brotherhood
28.2k members • Free
TJ’s PepSpace and WL Community
135 members • Free
Research Radar
11.8k members • $10/month
THE OPTIMIZATION HUB
222 members • Free
681 contributions to Orion Peptides
6 Lesser-Known Peptides Being Studied for Lifespan and Aging Pathways
I cannot do what I do without the support of Orion Peptides, whose commitment to research and education helps make content like this possible. Use code Parker15 for 15% off. When most people think about longevity research, they immediately think of compounds like NAD+, rapamycin, metformin, or GLP-1 medications. But behind the scenes, researchers are investigating an entirely different class of compounds that may influence some of the most important biological mechanisms associated with aging. Many of these peptides are not household names. In fact, most people have never heard of them. Yet they are being studied because they target pathways linked to mitochondrial function, cellular repair, inflammation, immune aging, stress resistance, and even telomere biology. Here are six of the most fascinating peptides currently being explored in longevity and healthy aging research. 1. SS-31 (Elamipretide) One of the most exciting areas of longevity science involves the mitochondria. Often referred to as the "powerhouses of the cell," mitochondria are responsible for producing the energy required for virtually every biological process. Unfortunately, mitochondrial function tends to decline with age. This is where SS-31 enters the conversation. SS-31, also known as Elamipretide, is a mitochondria-targeting peptide designed to interact directly with mitochondrial membranes. Researchers are studying it because it may: • support mitochondrial integrity • improve ATP production • reduce oxidative stress • preserve cellular energy production • support tissue repair mechanisms Why does this matter? Mitochondrial dysfunction is considered one of the major hallmarks of aging. When cellular energy production declines, organs and tissues often lose their ability to repair and regenerate efficiently. Current research is exploring SS-31 in relation to age-related changes affecting the heart, kidneys, muscles, and nervous system. 2. Klotho Peptides
1
0

If You Are Using Peptides, These Are the Labs You Should Actually Be Monitoring
I cannot do what I do without the support of Orion Peptides and the educational content they help make possible. Use code Parker15 for 15% off. One of the most overlooked parts of peptide use in research settings is not the compounds themselves, but the monitoring that should go alongside them. Peptides can have meaningful biological effects, but like any active compound interacting with human physiology, they can also influence different systems in the body. That’s why understanding lab work is just as important as understanding mechanisms. The goal is simple: Safety first, optimisation second. Why Lab Monitoring Matters Any compound that influences signalling pathways, metabolism, or tissue repair can potentially affect: • kidney function • liver enzymes • blood counts • inflammatory markers • hormonal pathways Even when compounds are well tolerated, biological systems still adapt and respond. Monitoring helps ensure those adaptations stay within healthy ranges. Growth Hormone (GH) Peptides For peptides that influence growth hormone signalling, such as GH secretagogues: Key marker: • IGF-1 levels IGF-1 provides a useful indirect indicator of GH activity and downstream metabolic signalling. Tracking this helps understand how strongly the pathway is being activated. NAD+ Related Protocols For NAD+ related compounds or metabolic support protocols: Key markers: • fasting glucose • liver function tests (ALT, AST) These markers help assess metabolic balance and hepatic processing over time. BPC-157 For peptides studied in tissue repair and inflammation modulation: Key markers: • kidney function (creatinine, eGFR) • complete blood count (CBC) These help monitor systemic response and general physiological stability. GHK-Cu For copper-based peptides involved in tissue regeneration pathways: Key markers: • liver function tests • serum copper levels • CRP (C-reactive protein) These provide insight into both metabolic handling and inflammatory status.
1
0

“You Need to Cycle Off Peptides” — The Myth That Doesn’t Match the Science
I cannot do what I do without the support of Orion Peptides and the educational content they help make possible. Use code Parker15 for 15% off. One of the most repeated claims in the peptide space is this: “You need to cycle off peptides.” It gets said so often that most people assume it’s established science. But when you actually look at the research, the idea doesn’t come from peptide biology at all. It comes from steroid biology. And those are two completely different systems. Where the Cycling Idea Actually Comes From Cycling makes sense in the context of anabolic steroids. When exogenous testosterone is introduced, the body detects elevated hormone levels. The pituitary reduces or shuts down natural production. That’s why steroid users cycle: • to allow endogenous hormone production to recover• to reduce long-term suppression• to manage hormonal balance So the idea of “cycling” is rooted in hormone suppression physiology. The key point: That mechanism applies to steroids, not peptides. Why Peptides Work Differently Peptides generally do not replace hormones in the body. Instead, they act as signalling molecules. They communicate with existing systems and encourage biological activity. They do not typically override endocrine production in the same way exogenous hormones do. That difference matters. GH Secretagogues: A Different Mechanism Entirely Compounds like: • CJC-1295 • Ipamorelin work by stimulating the pituitary gland to release growth hormone. They do not supply growth hormone externally. They signal your body to produce its own. Research on GH secretagogues has not shown the same type of shutdown pattern seen with exogenous hormone use. That is a fundamental distinction. Repair Peptides and Signalling Molecules Other commonly discussed peptides such as: • BPC-157 • TB-500 are studied primarily as signalling and repair-related molecules. Current research does not describe classic endocrine suppression pathways for these compounds.

Syringes on Amazon Are Next – Stock Up
I cannot do what I do without the support of Orion Peptides and the educational content they help make possible. Use code Parker15 for 15% off. There has been growing discussion around changes in how basic medical and research supplies are being distributed through large online marketplaces. Items such as syringes, bacteriostatic water, and related laboratory accessories have historically been widely available through e-commerce platforms, including Amazon and other large retailers. However, over the past few years, there has been a noticeable shift in how these products are listed, regulated, and distributed. This article breaks down what is actually happening from a systems perspective, without speculation or fear-based framing. Why These Products Are Under More Scrutiny Syringes and injectable-related supplies fall into a category that intersects: • medical use • laboratory use • harm reduction contexts • and controlled distribution frameworks in certain jurisdictions Because of this overlap, they are often subject to: • platform-level policy changes • regional regulatory requirements • seller verification standards • and restrictions on certain listings or keywords Large marketplaces periodically update their policies to comply with evolving legal and regulatory frameworks. This is not unique to one product category, but syringes and sterile consumables are often included due to their broad usage profile. The Role of Large E-Commerce Platforms Platforms like Amazon operate under strict compliance frameworks. This means they must balance: • consumer access • regulatory compliance • liability management • and seller verification standards As a result, certain medical-adjacent products may be: • reclassified • restricted to verified sellers • removed from general listings • or moved into more controlled categories These changes can appear sudden to consumers, but they are typically the result of gradual policy updates.
0
0

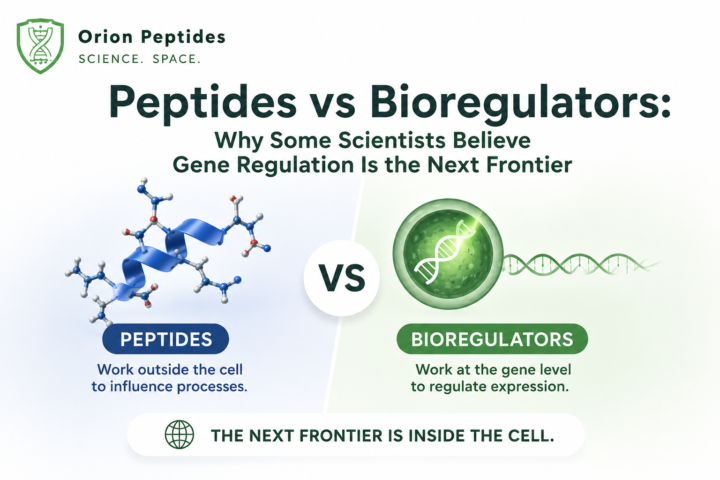
Peptides vs Bioregulators: Why Some Scientists Believe Gene Regulation Is the Next Frontier
I cannot do what I do without the support of Orion Peptides and the educational content they help make possible. Use code Parker15 for 15% off. Most people lump peptides and bioregulators into the same category. That is understandable. Both involve short chains of amino acids. Both are studied in longevity and regenerative medicine circles. Both are often discussed together in anti-aging conversations. But according to researchers studying these compounds, they may operate at very different levels of biology. The simplest way to understand it is this: Most peptides communicate with cells. Bioregulators may communicate with genes. That is a very different conversation. How Traditional Peptides Work Most peptides work by interacting with receptors found on the surface of cells. Think of a receptor as a lock. The peptide acts like a key. When the key fits, the cell receives a message and responds. Researchers have studied this mechanism extensively with compounds such as: • BPC-157 • TB-500 • CJC-1295 • Growth hormone secretagogues • GLP-1 related peptides The peptide binds to a receptor. The receptor activates a signaling pathway. The cell changes its behavior. This process is called cellular signaling. It is one of the primary ways biology communicates. The important point is that the signal originates outside the cell. What Makes Bioregulators Different? Bioregulators are much smaller molecules. Many consist of only 2 to 4 amino acids. Because of their size, researchers believe some bioregulators may be capable of crossing the cell membrane and entering the nucleus itself. Why does that matter? Because the nucleus contains DNA. And DNA controls how cells function. Rather than simply activating a receptor on the surface of the cell, bioregulators are being studied for their ability to influence gene expression directly. In simple terms: A traditional peptide may tell a cell what to do. A bioregulator may influence how the cell reads its own instruction manual.
1
0
1-10 of 681
Online now
Joined Dec 12, 2025

