Activity
Mon
Wed
Fri
Sun
Jun
Jul
Aug
Sep
Oct
Nov
Dec
Jan
Feb
Mar
Apr
What is this?
Less
More
Memberships
Josh's Chemistry Friends
11 members • Free
1 contribution to Josh's Chemistry Friends
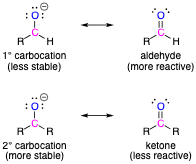
Difference between "less stable" and "more reactive"?
I thought "less stable" and "more reactive" meant the same thing, so I'm confused why the attached image from LibreTexts draws a distinction between them. Don't they both imply that a molecule is in a high energy state and liable to change in order to fix that?
1-1 of 1

